20220404 cell adhesion
From mn.fysikk.laglivlab
Followed the procedure
Contents
Assembling the chip
- Clean glass slide with IP and N2
- Punch holes for fluid inlets
- Used 1/16" punch to create the holes
- Place both PDMS and glass slide with clean sides up on glass plate in plasma cleaner
- Follow instructions for Plasma treatment
- Placed the PDMS on top of the glass slide and watch it attach
- Some pressure needed to be applied to bond
Filling the chip
For sterilizing and filling the chip with water and fibronectin you need
- 1ml syringe with
- 20G needle and
- 2cm long 1/16" teflon tube on the needle.
- small beaker
- ethanol
- distilled water
- Eppendorf tube of fibronectin
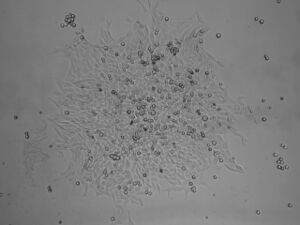
- Cells in fresh media
- Inlet and outlet tubes
- Measure lengths of inlet and outlet tubes and cut
- Sterilize the tubes
Sterilizing
No sterilizing today, everything was done in 420 instead of in 431
Water filling
- Fill a small amount of distilled water in beaker
- Retract <1ml distilled water into tube/needle/syringe
- Fit in inlet of chip and fill slowly
- Needle into tube first, then tube into inlet
- Empty syringe and needle to be ready to use for fibronectin
Fibronectin filling
- Take one Eppendorf with 0.5 ml fibronectin form freezer
- Thaw the fibronectin
- Retract fibronectin into tube/needle/syringe
- fit in inlet of chip and fill slowly
- Attach inlet and outlet tubes
- Incubate for 30 min in incubator
- Rinse with PBS
- Details?
- Pour ~?? ml PBS in sterile beaker
- Fill sterile ?? ml syringe+needle with PBS
- Slowly flow PBS through chip