Difference between revisions of "Microfluidics"
From mn.fysikk.laglivlab
Dagkd@uio.no (talk | contribs) (→In Giæver lab 420) |
Dagkd@uio.no (talk | contribs) |
||
| Line 129: | Line 129: | ||
* Stop all flow until cells have adhered to surface | * Stop all flow until cells have adhered to surface | ||
* Start slow flow of media through chip | * Start slow flow of media through chip | ||
| + | |||
| + | = Controlling O2 and CO2 concentration during experiment = | ||
| + | This is still on the planning stage. Check out [https://wiki.uio.no/mn/fysikk/laglivlab/index.php/Controlling_O2_and_CO2_level planning notes here]]. | ||
| + | |||
| + | = Measuring CO2 in the cell media = | ||
| + | Here we have not started the planning notes. Testing the electronics is the next step. | ||
Revision as of 15:25, 6 April 2022
Contents
Designing Microfluidic masks
- Stanford microfluidics foundry has a good guide for designing your own device. That includes a guide to use AutoCAD.
- AutoCAD (a program from Autodesk) is available free.
- You can use Klayout but we have more support for using Autocad.
- Mask template for placing your designs
- This guide for designing masks has some important tips
- All fluid pathways have to be inside one or more closed contour(s)
- The outer contour should be drawn in one layer (Give it a name like "Flow")
- Any obstacles inside this outer contour must be drawn in another layer. Give it a name like "Flow inner polygons")
- Displaying your design as PDF or otherwise is not straight forward because the resolution needed. You get a fair impression using CloudConvert which is much better than AutoCads own pdf export.
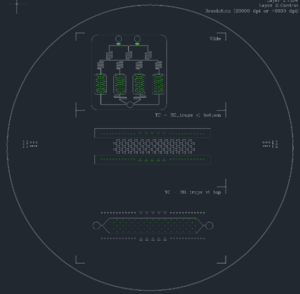
- Example of a design where white lines (in screen shot below, black lines in PDF) are contours containing flow and green lines are inner polygons. Here is a PDF version of the file and the DWG file.
- Rounding corners on a region
- Command EXPLODE makes region into lines
- Command JOIN joins lines into polylines
- Command FILLET, downarrow to get options RADIUS and POLYLINE
Photolithography
PDMS casting
- PDMS preparation
- Bring cup with PDMS close to wafer in Petri dish
- Pour slowly and avoid introducing air bubbles
- Remove air bubbles by puncturing them
- Fill petri dish to 5-10 mm above wafer surface
Assembling the chip
- Open Wiki lab notebook on computer to make notes of observations and deviations from this protocol
- Clean glass slide with IP and N2
- Punch holes for fluid inlets
- For 1/16" tubes use OD?? punch
- For thin teflon tube use OD?? punch
- Place both PDMS and glass slide with clean sides up on glass plate in plasma cleaner
- Follow instructions for Plasma treatment
- Place the PDMS on top of the glass slide and watch it attach
- If necessary, apply a gentle pressure to the PDMS
Filling the chip
- Open Wiki lab notebook on computer to make notes of observations and deviations from this protocol
For sterilizing and filling the chip with water and fibronectin you need
- 1ml syringe with
- 20G needle and
- 2cm long 1/16" teflon tube on the needle.
- small beaker
- ethanol
- distilled water
- Eppendorf tube of fibronectin
- Cells in fresh media
- Inlet and outlet tubes
- Measure lengths of inlet and outlet tubes and cut
- Sterilize the tubes
Sterilizing
- If you want a sterile chip move each object into LAF bench once they have been sterilized
- Spray chip with ethanol and rub off glass side
- Assemble syringe, needle and teflon tube
- Spray ethanol and rub outside
- Rinse beaker in ethanol and pour in a small amount of ethanol
- Retract syringe half way in air, and fill teflon tube with ethanol
- Empty syringe of ethanol and air, make sure it is dry
- Empty beaker of ethanol
Water filling
- Fill a small amount of distilled water in beaker
- Retract <1ml distilled water into tube/needle/syringe
- Fit in inlet of chip and fill slowly
- Empty syringe and needle to be ready to use for fibronectin
Fibronectin filling
- Take one Eppendorf with 0.5 ml fibronectin form freezer
- Thaw the fibronectin
- Retract fibronectin into tube/needle/syringe
- fit in inlet of chip and fill slowly
- Attach inlet and outlet tubes
- Incubate for 30 min in incubator
- Rinse with PBS
- Pour a few ml PBS in sterile beaker
- Fill sterile 1 ml syringe+needle with PBS
- Fit correct length of (sterile) inlet tube and outlet tube to chip
- Slowly flow PBS through chip
Cell filling
In cell lab 431
- Open Wiki lab notebook on computer to make notes of observations and deviations from this protocol
- Follow protocol for [cell passaging]
- Transfer the some of cell suspension to a sterile centrifuge tube of appropriate size (Eppendorf or other depending on volume)
- Centrifuge for 10 minutes at 800 × g.
- Note: Certain cell lines are very sensitive to centrifugal force.
- Carefully remove the supernatant without disturbing the cell pellet.
- Add the desired volume of fresh medium gently to the side of the tube and slowly pipette up and down 2 to 3 times to resuspend the cell pellet.
- Transfer the cells to a sterile syringe or sample holder
- Fill another syringe or sample holder with fresh media
In Giaver lab 420
You now need to have
- Wiki lab notebook open to make notes of observations and deviations from this protocol
- Computer with microscope and camera ready
- turn on computer, microscope and start flycap
- test imaging on an old chip (start with 4x, turning the objective turret CCW: 4X - 10X - 20X - 40X)
- 2 syringe pumps or pressure controller ready
- Syringe pumps: adjust syringe size and flow rates to 1-60 ul/min
- Pressure controller: set initial pressure to zero on both channels
- The sterile chip
- with fibronectin on the glass
- filled with PBS
- inlet and outlet tubes connected
- A syringe or sample holder with cells suspended in fresh media
- If possible: bubble correct air/CO2 mixture through media before starting
Now it's time to fit everything together on top of the microscope
- Place the chip on the microscope, fasten with gum and focus on area by inlet
- Connect syringe pumps or pressure regulator:
- Syringe pumps: Place syringes in syringe pumps and test that they engage
- Pressure regulator: Connect air tubes from regulator to sample holders
- Connect sample holders/syringes to the chip via the T-connector (see figure)
- Connect output tube to a waste container
- Press media through T-connector, chip and to cell syringe/sample holder until all air bubbles have been removed
- Stop flow from media container
Running experiment
- Start slow flow from cell container to chip
- Follow images. Stop flow now and then to check if cells have reached chip
- Start recording image sequence to D:\Microfluidic\2022...\
- Stop all flow until cells have adhered to surface
- Start slow flow of media through chip
Controlling O2 and CO2 concentration during experiment
This is still on the planning stage. Check out planning notes here].
Measuring CO2 in the cell media
Here we have not started the planning notes. Testing the electronics is the next step.